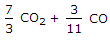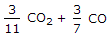Mechanical Engineering - Thermodynamics - Discussion
Discussion Forum : Thermodynamics - Section 6 (Q.No. 4)
4.
The mass of carbon per kg of flue gas is given by
Answer: Option
Explanation:
The mass of carbon, contained in 1 kg of flue or exhaust gases, may be calculated from the mass of carbon dioxide and carbon monoxide present in them.
We know that 1 kg of carbon produces 11/3 kg of carbon dioxide. Hence 1 kg of carbon dioxide will contain 3/11 kg of carbon. Also, 1 kg of carbon produces 7/3 kg of carbon monoxide, hence 1 kg of carbon monoxide will contain 3/7 kg of carbon.
Mass of carbon per kg of flue gas is given by:
3/11 CO2 + 3/7 CO.
Discussion:
25 comments Page 1 of 3.
Jagdish said:
3 years ago
I think it's 3/11 CO2, 3/7 CO.
(1)
Vinay said:
5 years ago
Mass of carbon per kg of flue gas is given by:
3/11 CO2 + 3/7 CO.
3/11 CO2 + 3/7 CO.
(1)
Sripati sahoo said:
6 years ago
I agree with @Mr. VelmuruganR. You are perfectly explained.
(1)
Velmurugan R said:
7 years ago
The mass of carbon, contained in 1 kg of flue or exhaust gases, may be calculated from the mass of carbon dioxide and carbon monoxide present in them.
We know that 1 kg of carbon produces 11 / 3 kg of carbon dioxide. Hence 1 kg of carbon dioxide will contain 3 / 11 kg of carbon. Also, 1 kg of carbon produces 7/3 kg of carbon monoxide, hence 1 kg of carbon monoxide will contain 3/7 kg of carbon.
Mass of carbon per kg of flue gas is given by:
3/11 CO2+ 3/7 CO.
We know that 1 kg of carbon produces 11 / 3 kg of carbon dioxide. Hence 1 kg of carbon dioxide will contain 3 / 11 kg of carbon. Also, 1 kg of carbon produces 7/3 kg of carbon monoxide, hence 1 kg of carbon monoxide will contain 3/7 kg of carbon.
Mass of carbon per kg of flue gas is given by:
3/11 CO2+ 3/7 CO.
(4)
KAPIL said:
7 years ago
I think the answer would be 3/11CO2 + 3/7CO.
Tejaa said:
8 years ago
Yes, right @Syed Sameer.
SYED SAMEER said:
8 years ago
The mass of carbon, contained in 1 kg of flue or exhaust gases, may be calculated from the mass of carbon dioxide and carbon monoxide present in them.
We know that 1 kg of carbon produces 11/3 kg of carbon dioxide. Hence 1 kg of carbon dioxide will contain 3/11 kg of carbon. Also, 1 kg of carbon produces 7/3 kg of carbon monoxide, hence 1 kg of carbon monoxide will contain 3/7 kg of carbon.
Mass of carbon per kg of flue gas is given by:
3/11CO2 + 3/7CO.
We know that 1 kg of carbon produces 11/3 kg of carbon dioxide. Hence 1 kg of carbon dioxide will contain 3/11 kg of carbon. Also, 1 kg of carbon produces 7/3 kg of carbon monoxide, hence 1 kg of carbon monoxide will contain 3/7 kg of carbon.
Mass of carbon per kg of flue gas is given by:
3/11CO2 + 3/7CO.
K K said:
8 years ago
@Biplab.
CO2 + CO = C is not balanced.
I think Balanced equation is CO2+2CO=3C+2O2.
CO2 + CO = C is not balanced.
I think Balanced equation is CO2+2CO=3C+2O2.
J.T.R said:
8 years ago
No, the correct answer is 3/11 co2+ 3/7 co.
Mehul said:
8 years ago
Right answer is 3/11CO2 + 3/7CO.
Post your comments here:
Quick links
Quantitative Aptitude
Verbal (English)
Reasoning
Programming
Interview
Placement Papers