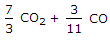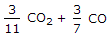Mechanical Engineering - Thermodynamics
Exercise : Thermodynamics - Section 6
1.
The atomic mass of sulphur is
2.
A path 1-2-3 is given. A system absorbs 100 kJ as heat and does 60 kJ of work while along the path 1-4-3, it does 20 kJ of work. The heat absorbed during the cycle 1-4-3 is


3.
The atomic mass of an element is the number of times the atom of that element is heavier than the hydrogen atom.
4.
The mass of carbon per kg of flue gas is given by
Answer: Option
Explanation:
The mass of carbon, contained in 1 kg of flue or exhaust gases, may be calculated from the mass of carbon dioxide and carbon monoxide present in them.
We know that 1 kg of carbon produces 11/3 kg of carbon dioxide. Hence 1 kg of carbon dioxide will contain 3/11 kg of carbon. Also, 1 kg of carbon produces 7/3 kg of carbon monoxide, hence 1 kg of carbon monoxide will contain 3/7 kg of carbon.
Mass of carbon per kg of flue gas is given by:
3/11 CO2 + 3/7 CO.
5.
A cycle consisting of two isothermal and two isentropic processes, is known as
Quick links
Quantitative Aptitude
Verbal (English)
Reasoning
Programming
Interview
Placement Papers