Electronics and Communication Engineering - Electronic Devices and Circuits - Discussion
Discussion Forum : Electronic Devices and Circuits - Section 3 (Q.No. 2)
2.
As per Einstein's equation, the velocity of emitted electron in photoelectric emission is given by the equation
Answer: Option
Explanation:
Some electrons have less energy than Fermi level E and, therefore, more energy than Uw to escape.
Therefore, this equation has inequality sign.
Discussion:
2 comments Page 1 of 1.
Tarif said:
2 months ago
Einstein's photoelectric equation states that the maximum kinetic energy (0.5*m*vmax^2) of an emitted electron is equal to the energy of the incident photon (hf) minus the work function (Uw) of the material
So: 0.5 * m * vmax^2 = hf - Uw
However, not all electrons absorb the photon energy at the surface; some absorb energy deeper within the material and lose some energy before escaping. Therefore, the kinetic energy of an electron upon emission can be any value up to the maximum kinetic energy. This is represented by the inequality given in options B & D.
So: 0.5 * m * vmax^2 = hf - Uw
However, not all electrons absorb the photon energy at the surface; some absorb energy deeper within the material and lose some energy before escaping. Therefore, the kinetic energy of an electron upon emission can be any value up to the maximum kinetic energy. This is represented by the inequality given in options B & D.
Jinli said:
6 years ago
Can someone explain this equation further?
(1)
Post your comments here:
Quick links
Quantitative Aptitude
Verbal (English)
Reasoning
Programming
Interview
Placement Papers
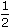 mv2 = hf - Uw
mv2 = hf - Uw