Chemical Engineering - Chemical Engineering Thermodynamics
Exercise : Chemical Engineering Thermodynamics - Section 11
- Chemical Engineering Thermodynamics - Section 1
- Chemical Engineering Thermodynamics - Section 2
- Chemical Engineering Thermodynamics - Section 3
- Chemical Engineering Thermodynamics - Section 4
- Chemical Engineering Thermodynamics - Section 5
- Chemical Engineering Thermodynamics - Section 6
- Chemical Engineering Thermodynamics - Section 7
- Chemical Engineering Thermodynamics - Section 8
- Chemical Engineering Thermodynamics - Section 9
- Chemical Engineering Thermodynamics - Section 10
- Chemical Engineering Thermodynamics - Section 11
36.
To obtain integrated form of Clausius-Clayperon equation, 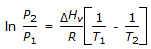 , from the exact Clayperon equafion, it is assumed that the
, from the exact Clayperon equafion, it is assumed that the
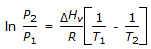 , from the exact Clayperon equafion, it is assumed that the
, from the exact Clayperon equafion, it is assumed that the37.
In case of an __________ process, the temperature of the system increases.
38.
The standard state of a gas (at a given temperature) is the state in which fugacity is equal to
39.
Cp of a gas at its critical temperature and pressure
40.
Trouton's ratio is given by (where λb, = molal heat of vaporisation of a substance at its normal boiling point, kcal/kmol Tb = normal boiling point, °K )
Quick links
Quantitative Aptitude
Verbal (English)
Reasoning
Programming
Interview
Placement Papers