Chemical Engineering - Chemical Engineering Thermodynamics
Exercise : Chemical Engineering Thermodynamics - Section 11
- Chemical Engineering Thermodynamics - Section 1
- Chemical Engineering Thermodynamics - Section 2
- Chemical Engineering Thermodynamics - Section 3
- Chemical Engineering Thermodynamics - Section 4
- Chemical Engineering Thermodynamics - Section 5
- Chemical Engineering Thermodynamics - Section 6
- Chemical Engineering Thermodynamics - Section 7
- Chemical Engineering Thermodynamics - Section 8
- Chemical Engineering Thermodynamics - Section 9
- Chemical Engineering Thermodynamics - Section 10
- Chemical Engineering Thermodynamics - Section 11
31.
The third law of thermodynamics states that the
32.
Consider the reaction, C + O2  CO2 ;ΔH = - 94 kcal. What will be the value of ΔH for the reaction CO2
CO2 ;ΔH = - 94 kcal. What will be the value of ΔH for the reaction CO2 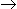 C + O2 ?
C + O2 ?
 CO2 ;ΔH = - 94 kcal. What will be the value of ΔH for the reaction CO2
CO2 ;ΔH = - 94 kcal. What will be the value of ΔH for the reaction CO2 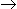 C + O2 ?
C + O2 ?33.
Which is not a state function ?
34.
For an ideal liquid solution, which of the following is unity ?
35.
In the decomposition of PCl5 represented by, PCl5  PCl3 + Cl2, decrease in the pressure of the system will __________ the degree of dissociation of PCl5.
PCl3 + Cl2, decrease in the pressure of the system will __________ the degree of dissociation of PCl5.
 PCl3 + Cl2, decrease in the pressure of the system will __________ the degree of dissociation of PCl5.
PCl3 + Cl2, decrease in the pressure of the system will __________ the degree of dissociation of PCl5.Quick links
Quantitative Aptitude
Verbal (English)
Reasoning
Programming
Interview
Placement Papers