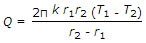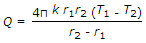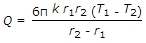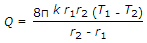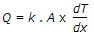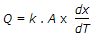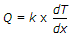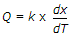Mechanical Engineering - Heat Transfer, Refrigeration and Air Conditioning
Exercise : Heat Transfer, Refrigeration and Air Conditioning - Section 1
- Heat Transfer, Refrigeration and Air Conditioning - Section 1
- Heat Transfer, Refrigeration and Air Conditioning - Section 2
- Heat Transfer, Refrigeration and Air Conditioning - Section 3
- Heat Transfer, Refrigeration and Air Conditioning - Section 4
- Heat Transfer, Refrigeration and Air Conditioning - Section 5
- Heat Transfer, Refrigeration and Air Conditioning - Section 6
- Heat Transfer, Refrigeration and Air Conditioning - Section 7
- Heat Transfer, Refrigeration and Air Conditioning - Section 8
11.
According to Dalton's law of partial pressures, (where pb = Barometric pressure, pa = Partial pressure of dry air, and pv = Partial pressure of water vapour)
12.
The heat transfer by conduction through a thick sphere is given by
13.
The relative humidity is defined as
14.
Fourier's law of heat conduction is (where Q = Amount of heat flow through the body in unit time, A = Surface area of heat flow, taken at right angles to the direction of heat flow, dT = Temperature difference on the two faces of the body, dx = Thickness of the body, through which the heat flows, taken along the direction of heat flow, and k = Thermal conductivity of the body)
15.
The heat transfer by conduction through a thick sphere is same as through a thick cylinder.
Quick links
Quantitative Aptitude
Verbal (English)
Reasoning
Programming
Interview
Placement Papers