Engineering Mechanics - PKRB: Force and Animation - Discussion
Discussion Forum : PKRB: Force and Animation - General Questions (Q.No. 3)
3.
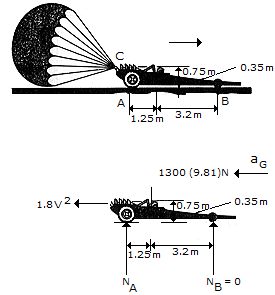
The dragster has a mass of 1.3 Mg and a center of mass at G. If a braking parachute is attached at C and provides a horizontal braking force FD, determine the maximum deceleration the dragster can have upon releasing the parachute without tipping the dragster over backwards (i.e., the normal force under the wheels and assume that the engine is disengaged so that the wheels are freely rolling.
Discussion:
Be the first person to comment on this question !
Post your comments here:
Quick links
Quantitative Aptitude
Verbal (English)
Reasoning
Programming
Interview
Placement Papers