Chemical Engineering - Chemical Reaction Engineering - Discussion
Discussion Forum : Chemical Reaction Engineering - Section 1 (Q.No. 17)
17.
Reaction rate equation for the reaction, 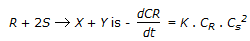 . If S at is present in large excess, what is the order of this reaction?
. If S at is present in large excess, what is the order of this reaction?
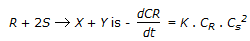 . If S at is present in large excess, what is the order of this reaction?
. If S at is present in large excess, what is the order of this reaction?Discussion:
3 comments Page 1 of 1.
Saurabh yadav said:
9 years ago
Because the reaction is elementary one, the rate determining element/compound is are i.e. why are power will decide the order of reaction?
(2)
Sankar Ghosh said:
1 decade ago
The limiting reactant becomes R, so the kinetics of the reaction depend only on the concentration of R.
(7)
Ashu said:
1 decade ago
Because concentration of S is too large therefore it can not show appreciable change in its concentration and we can include it in constant. Hence order of reaction is 1 with respect to A. This mechanism is called as Pseudo order reaction.
(9)
Post your comments here:
Quick links
Quantitative Aptitude
Verbal (English)
Reasoning
Programming
Interview
Placement Papers