Chemical Engineering - Chemical Reaction Engineering
Exercise : Chemical Reaction Engineering - Section 1
- Chemical Reaction Engineering - Section 1
- Chemical Reaction Engineering - Section 2
- Chemical Reaction Engineering - Section 3
- Chemical Reaction Engineering - Section 4
- Chemical Reaction Engineering - Section 5
- Chemical Reaction Engineering - Section 6
- Chemical Reaction Engineering - Section 7
- Chemical Reaction Engineering - Section 8
- Chemical Reaction Engineering - Section 9
- Chemical Reaction Engineering - Section 10
31.
The increase in the rate of reaction with temperature is due to
32.
A catalyst loses its activity due to
33.
'N' plug flow reactors in series with a total volume 'V' gives the same conversion as a single plug flow reactor of volume 'V' for __________ order reactions.
34.
Specific rate constant for a second order reaction
35.
For the irreversible elementary reactions in parallel viz 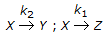 , the rate of disappearance of 'X' is equal to
, the rate of disappearance of 'X' is equal to
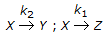 , the rate of disappearance of 'X' is equal to
, the rate of disappearance of 'X' is equal toQuick links
Quantitative Aptitude
Verbal (English)
Reasoning
Programming
Interview
Placement Papers