Chemical Engineering - Chemical Engineering Thermodynamics - Discussion
Discussion Forum : Chemical Engineering Thermodynamics - Section 2 (Q.No. 32)
32.
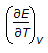 is the mathematical expression for
is the mathematical expression forDiscussion:
11 comments Page 1 of 2.
Vali said:
1 decade ago
This is mathematical expression for heat capacity at constant volume. So the answer is A.
Dare said:
1 decade ago
It's an expression for CV. A is the correct answer.
(1)
Chemi said:
10 years ago
Yes, the answer is A.
Maniteja said:
9 years ago
The answer is A.
Akash Varma said:
8 years ago
Absolutely its A.
E is the Internal Energy.
E is the Internal Energy.
(1)
Anooj pillai said:
6 years ago
Answer is A.
Rajput said:
5 years ago
We use (U) for denoting internal energy. But here E is not denoting internal energy. So, A can't be the right answer.
(1)
Naincy said:
5 years ago
The correct Answer should be A.
(1)
TANMAY SHAH said:
3 years ago
Here, Cv = (dU/dT)v.
(1)
Ramsajan said:
3 years ago
Cv=(dE/dT)v
So, Option (A) is right.
So, Option (A) is right.
(1)
Post your comments here:
Quick links
Quantitative Aptitude
Verbal (English)
Reasoning
Programming
Interview
Placement Papers