Chemical Engineering - Chemical Engineering Thermodynamics
Exercise : Chemical Engineering Thermodynamics - Section 11
- Chemical Engineering Thermodynamics - Section 1
- Chemical Engineering Thermodynamics - Section 2
- Chemical Engineering Thermodynamics - Section 3
- Chemical Engineering Thermodynamics - Section 4
- Chemical Engineering Thermodynamics - Section 5
- Chemical Engineering Thermodynamics - Section 6
- Chemical Engineering Thermodynamics - Section 7
- Chemical Engineering Thermodynamics - Section 8
- Chemical Engineering Thermodynamics - Section 9
- Chemical Engineering Thermodynamics - Section 10
- Chemical Engineering Thermodynamics - Section 11
11.
The variation of heat of reaction with temperature at constant pressure is given by the __________ law.
12.
At absolute zero temperature, the __________ of the gas is zero.
13.
Which of the following equations is used for the prediction of activity co-efficient from experiments ?
14.
The equation, 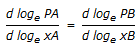 , applicable to a binary solution of components. A and B in equilibrium with their vapors at constant temperature and pressure is called the __________ equation.
, applicable to a binary solution of components. A and B in equilibrium with their vapors at constant temperature and pressure is called the __________ equation.
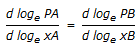 , applicable to a binary solution of components. A and B in equilibrium with their vapors at constant temperature and pressure is called the __________ equation.
, applicable to a binary solution of components. A and B in equilibrium with their vapors at constant temperature and pressure is called the __________ equation.15.
Entropy of a substance remains constant during a/an __________ change.
Quick links
Quantitative Aptitude
Verbal (English)
Reasoning
Programming
Interview
Placement Papers