Chemical Engineering - Chemical Reaction Engineering
Exercise : Chemical Reaction Engineering - Section 9
- Chemical Reaction Engineering - Section 1
- Chemical Reaction Engineering - Section 2
- Chemical Reaction Engineering - Section 3
- Chemical Reaction Engineering - Section 4
- Chemical Reaction Engineering - Section 5
- Chemical Reaction Engineering - Section 6
- Chemical Reaction Engineering - Section 7
- Chemical Reaction Engineering - Section 8
- Chemical Reaction Engineering - Section 9
- Chemical Reaction Engineering - Section 10
36.
With increase in K2/K1 in case of a unimolecular type elementary reactions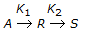 , the fractional yield of 'R' in mixed reactor (for a given conversion of 'A')
, the fractional yield of 'R' in mixed reactor (for a given conversion of 'A')
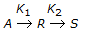 , the fractional yield of 'R' in mixed reactor (for a given conversion of 'A')
, the fractional yield of 'R' in mixed reactor (for a given conversion of 'A')37.
Collision theory gives the rate constant for bimolecular reaction as
38.
Find a mechanism that is consistent with the rate equation and reaction given below:
2A + B A2B, ( - rA) = k.CA.CB
A2B, ( - rA) = k.CA.CB
2A + B
 A2B, ( - rA) = k.CA.CB
A2B, ( - rA) = k.CA.CB39.
A gaseous reactant is introduced in a mixed reactor of 3 litres volume at the rate of 1 litre/second. The space time is __________ seconds.
40.
The temperature dependence of reaction rate constant (K) by Arhenius law is given by
Quick links
Quantitative Aptitude
Verbal (English)
Reasoning
Programming
Interview
Placement Papers
 AB ; AB + A
AB ; AB + A